Polybutadiene (PB) and acrylonitrile butadiene styrene (ABS) are widely used polymers in a number of products (including vehicle tires, kitchen appliances, computer hardware, toys and others), but are not generally recycled. Researchers at Oak Ridge National Laboratory (ORNL; Oak Ridge, Tenn.; www.ornl.gov) recently reported on a method that utilizes this difficult-to-recycle waste in making a range of high-quality materials.
The ORNL scientists developed a method for using waste PB and ABS as drop-in additives for the synthesis of other industrially important co-polymers. Using a ruthenium catalyst (2nd-generation Grubbs catalyst), the team was able to cleave the polymer chain of the waste polymer at specific points where a carbon-carbon double bond exists in the PB and ABS backbone and incorporate the waste plastic into a newly forming co-polymer.
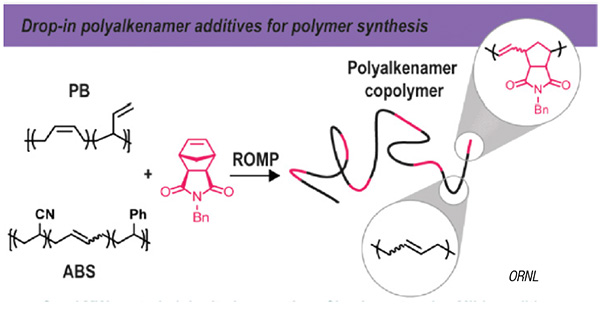
To do this, the scientists employed two types of metathesis reactions, in which double bonds break and form between carbon atoms, allowing polymer subunits to swap. In one case, a ring-opening metathesis polymerization (ROMP) occurs, and in another case, a cross-metathesis reaction occurs, incorporating the waste plastic into upcycled material.
“The Ru catalyst serves two functions,” explains ORNL scientist Jeff Foster, “to open the C=C bonds of cyclic alkene monomers and elongate the growing polymer chains, and to scramble the growing polymer chains with the waste PB or ABS. The outcome is a sequence comprised of both the monomers and the waste plastic.”
The molecular weight and composition of the upcycled polymer can be controlled by the amount of waste (PB or ABS) and monomer that are added to the reactor. “Generally, adding more monomer increases the molecular weight, the stiffness of the material and its softening temperature,” Foster notes. A more difficult challenge is controlling where the backbone insertion occurs. “That depends on the rate at which the monomer is ring-opened relative to scrambling, and how susceptible the C=C bonds derived from the monomer are to further reaction,” Foster says. “We are currently developing strategies to precisely control where insertion happens.”
The ORNL polymer insertion method is highly atom-economical and conserves most of the waste plastic matter that is added, Foster says. The ORNL team is exploring how to extend the technology to other waste streams with chemical functionality in their polymer backbone, such as styrene-butadiene rubbers (the major synthetic component of vehicle tires).
The research appears in a recent issue of the Journal of the American Chemical Society.