Concerns over limited supplies of petroleum-based feedstocks, coupled with the global issue of climate change, have increased demand from consumers and companies for more sustainable products. In this economic environment, chemical and polymer production from renewable resources is increasingly viewed as an attractive area for investment. Following this global trend, research on the production of 2,5-furandicarboxyllic acid (FDCA) has intensified over the last few years. FDCA has the potential to replace fossil-based purified terephthalic acid (PTA) in the production of polyesters and other polymers containing an aromatic ring. The most promising application segment for FDCA is for the production of polyethylene furanoate (PEF). This polymer exhibits physical properties and applications similar to polyethylene terephthalate (PET), and is produced by the use of FDCA, instead of PTA, in the PET-production process.
Production process
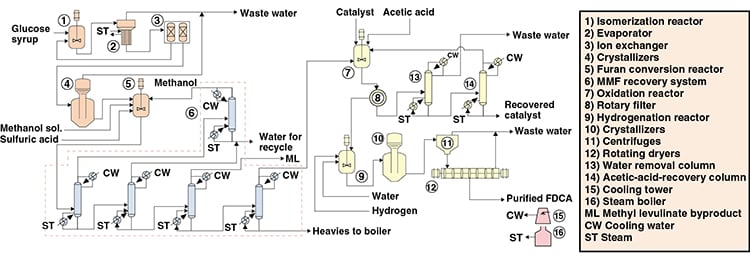
Figure 1 depicts an FDCA production process from a glucose solution via the furan pathway in a process similar to that developed by Avantium Technologies B.V. (Amsterdam, the Netherlands; www.avantium.com). This process can be divided into three main areas: furans production, methoxy methyl furan (MMF) purification, and FDCA production.
Furans production. The glucose content of the feed is first enzymatically isomerized to fructose, and the effluent is passed through ion-exchange resins to separate unreacted glucose from the fructose. The former is recycled to the isomerization reactor and the latter is sent to the furans conversion reactor. In the furans conversion reactor, crystallized fructose is solubilized with a 95 wt.% methanol aqueous solution and sulfuric acid. The reaction is conducted at 50 bars and 200ºC. Fructose is dehydrated to hydroxyl methyl furan (HMF) and, due to the excess of methanol, most HMF is converted to MMF, which is more stable than HMF. MMF purification. The reactor effluent is then sent to a distillation system to recover the solvent and separate the MMF from other impurities. Methanol and water are recovered and returned to the furans conversion area, while methyl levulinate is recovered as a byproduct. Also, a heavies stream is recovered to be used as boiler fuel, and finally, purified MMF is recovered and directed to the FDCA production area. FDCA production. Purified MMF is mixed with acetic acid solvent and a catalyst in the oxidation reactor to be converted to FDCA. This stream is filtered to recover crude FDCA. The acetic-acid-rich liquid is purified using two distillation columns. Catalyst is also recovered, but it must be reactivated before being recycled to the process. Crude FDCA is mixed with water and hydrogen in a hydrogenation reactor, where impurities are removed. The effluent from the reactor is crystallized, and purified FDCA is recovered from the solid materials. The liquid effluent is a waste stream.
Economic performance
An economic evaluation of an FDCA plant was conducted, assuming a facility with a nominal capacity of 300,000 ton/yr of purified FDCA constructed on the U.S. Gulf Coast. Included was a storage capacity equal to 20 days of operation for feedstocks and products. Estimated capital expenses (total fixed investment, working capital and initial expenses) to construct the plant are about $600 million, while the operating expenses are estimated at about $1,550/ton of purified FDCA.
Global perspective
The main production cost, as in many commodities-production processes, is the purchase of raw materials. Not only is glucose syrup an expensive source of glucose for this process, but also the conversion of fructose to MMF is relatively low, increasing the raw-material consumption. It would be beneficial for the profitability of the process to integrate it with biomass treatment, in order to obtain raw materials at lower costs. This process could easily integrate with a biorefinery, where it is possible to achieve a more efficient valorization of the biomass used as feedstock. Editor’s Note: The content for this column is supplied by Intratec Solutions LLC (Houston; www.intratec.us) and edited by Chemical Engineering. The analyses and models presented are prepared on the basis of publicly available and non-confidential information. The content represents the opinions of Intratec only. More information about the methodology for preparing analysis can be found, along with terms of use, at www.intratec.us/che.