Physical adsorption phenomena play a critical role in many separation and purification processes, including separations of industrial gases, purification of gases, moisture removal from fluids, water purification and others. Adsorption is a surface phenomenon in which a fluid (gas or liquid) molecule attaches to the surface of a material by physical forces known as Van der Waals interactions. This one-page reference discusses the nature of Van der Waals forces, and their role in adsorption processes, with a focus on London dispersion forces.
Van der Waals forces
Van der Waals forces is a general term that describes the relatively weak (compared to typical covalent bond strengths) intermolecular forces that result from individual electrostatic attractions between otherwise non-attracting atoms and molecules. Van der Waals interactions can be classified into three categories, as follows:
Hydrogen bonds. Hydrogen bonds form between H atoms — that acquire significant positive charge from being bound to electronegative elements — and negatively charged lone electron pairs on adjacent molecules. The most important H-bonds form with hydroxide and amide groups. Hydrogen bonds have strengths ranging from 5 to 50 kJ/mol.
Dipole-dipole interactions. Dipole-dipole interactions are the attractive forces that arise when the partial positive and negative charges that form on polar molecules are attracted to each other. These permanent attractive forces act at close range.
London dispersion forces. London dispersion forces refer to the electrostatic attraction of spontaneously formed transient temporary dipoles that arise in non-polar molecules. For adsorption phenomena, London forces are the most important.
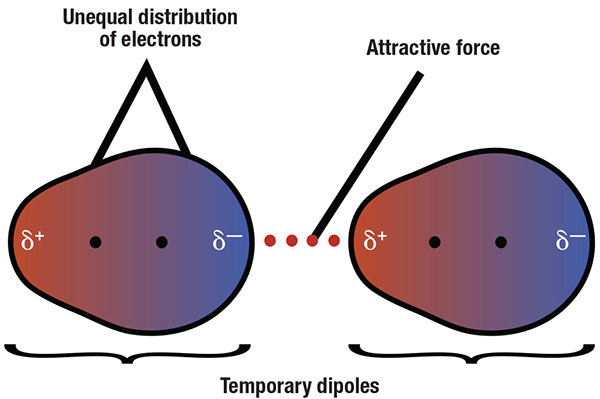
Figure 1. Transient dipoles resulting from spontaneous shifts in electron densities among molecules give rise to attractive forces
London forces
For a neutral molecule or atom, the average charge distribution is zero, but at any given instant, molecules have asymmetric distributions of electron density. Temporarily fluctuating dipole moments can arise from a brief shift of orbital electrons to one side of an atom or molecule. In adsorption, these transient dipoles induce complementary dipoles in neighboring molecules so that adsorbents are attracted to adsorbate molecules and they attach. These temporary induced dipoles are known as London dispersion forces, named for Fritz London, the German physicist and Duke University professor who described the intermolecular forces of noble gas atoms in 1930.
London forces have the following characteristics:
Non-chemical. London forces attract molecules to each other, but do not chemically change the molecules
Additive. The observed London force is the sum of all the individual interactions of the adsorbate molecule and the neighboring molecules. In adsorption onto activated carbon, for example, the magnitude of the London forces will be related to the density of carbon within the vicinity of the adsorbate molecules
Nonspecific. London forces are present among all molecules
Temperature-independent. London forces are unaffected by temperature, and thus the adsorption forces will be constant with temperature. Adsorption capacities will still be sensitive to the changes in vapor pressure or solubility of the adsorbing molecules that result from temperature differences
Short-ranged. The magnitude of the London force is sensitive to the proximity of the adsorbate molecules from the adsorbent material. London forces can be considered negligible at separations greater than about two molecular layers. In activated carbon, the adsorption forces are significant if the gaps or voids within the pores of the carbon structure (pore widths) are less than four or five molecular layers.
Bond strengths
Bonding energies in adsorption range from about 10 to 70 kJ/mol, much lower than typical covalent bond energies of 200 to 500 kJ/mol. The adsorption bonding energy is high enough for adsorption to occur, yet low enough to allow the adsorbent to be regenerated by removing the adsorbed molecules.
Adsorption
The affinity of a fluid component for a particular adsorbent depends on molecular characteristics, such as size, shape, and polarity of the adsorbent material, as well as the partial pressure or concentration in the fluid, and the system temperature.
Commercial adsorbents are highly porous, with pore surface areas ranging from about 100 to 1,200 m2/g. The large surface area allows a large amount of adsorption relative to the weight of the adsorbent. A number of factors can affect adsorption, such as pore size distribution, molecular size of the impurity species, particle size of the adsorbent, pH of the solution and others. Activated carbon is one of the most complex solid adsorbents, and is very versatile because of its extremely high surface area and micropore volume.
Department Editor: Scott Jenkins
Editor’s note: Information for this column came from several sources, including: Clark, J., Intermolecular Bonding, article in Chemguide (www.chemguide.co.uk) 2017; Desotech, Carbonology articles: What is adsorption?, www.desotech.com, accessed June 2021; and others